Conservation of Energy Simulator
Explore the principle of conservation of energy by varying conditions and observing how energy transforms and transfers in an interactive simulator.
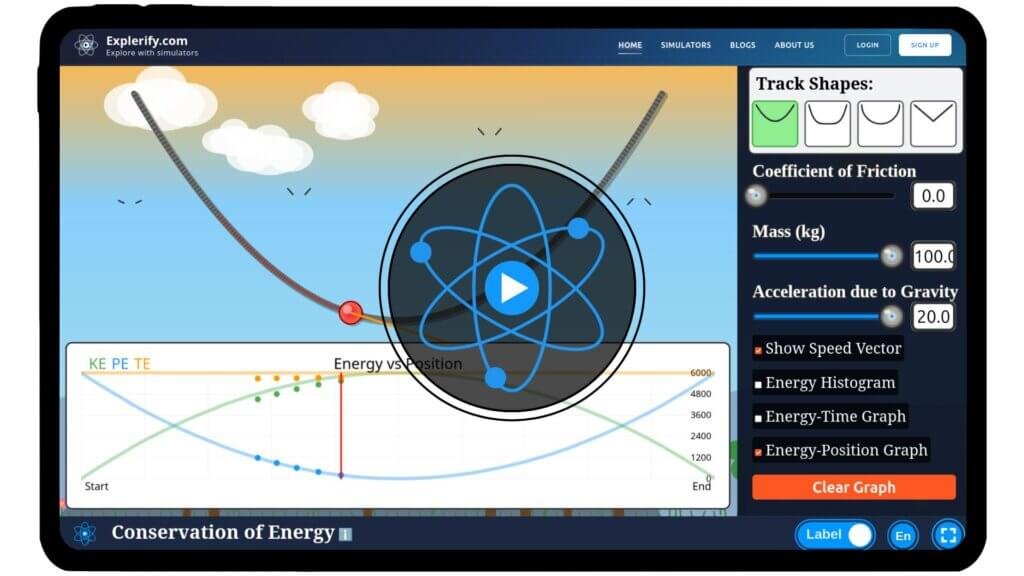
Conservation of Energy
Physics surrounds us, influencing countless aspects of our daily lives, often in ways we overlook. Have you ever wondered how energy changes form while never being lost? From a swinging pendulum to a rolling object, energy is constantly transferred and transformed. With our interactive simulator, you can explore the law of conservation of energy firsthand. Adjust system parameters, observe energy transformations in real time, and uncover the principles that govern motion and energy transfer. Start your journey into the fascinating world of physics — experiment with conservation of energy today!
\(E = K + U = \frac{1}{2}mv^2 + mgh \)
Mathematical description
where:
- \( E \) is the total mechanical energy.
- \( m \) is the mass of the ball
- \( v \) is the velocity of the ball.
- \( g \) is the acceleration due to gravity.
- \( h \) is the vertical height of the ball from the reference level.
FAQs
Qus 1. What is the law of conservation of energy?
The Law of Conservation of Energy states that energy can neither be created nor destroyed. It can only be transformed from one form to another. The total energy of an isolated system always remains constant. This principle applies to all physical and chemical processes
Qus 2. What are some examples of conservation of energy in daily life?
When a ball falls, its potential energy converts into kinetic energy. In electric bulbs, electrical energy transforms into light and heat energy. In vehicles, chemical energy from fuel changes into mechanical energy. In all cases, total energy remains conserved.
Qus 3. How does energy change form while remaining conserved?
Energy can transform between forms such as kinetic, potential, thermal, chemical, and electrical energy, but the total energy of a closed system remains constant.
Qus 4. What is an isolated system in terms of energy?
An isolated system is one where no energy enters or leaves the system. Since there is no external interaction, the total energy within the system remains constant. The law of conservation of energy strictly applies to such systems. Real-world systems often approximate isolation.
Qus 5. How does conservation of energy apply to mechanical systems?
In mechanical systems, energy usually shifts between kinetic and potential energy. For example, a swinging pendulum continuously converts energy between these two forms. If no energy is lost to friction or air resistance, the total mechanical energy remains const
Qus 6. Is energy conserved when friction is present?
Yes, energy is still conserved even when friction acts. However, some mechanical energy converts into heat and sound. While useful energy decreases, the total energy of the system and surroundings remains constant. Energy is not lost, only transformed.
Qus 7. What is the historical significance of the conservation of energy law?
The Law of Conservation of Energy is historically significant because it unified heat, work, and motion under a single scientific principle. In the 19th century, experiments by scientists like James Prescott Joule proved that different forms of energy are interchangeable.
This shifted physics away from isolated force-based explanations to energy-based thinking. The law became a cornerstone for modern physics, engineering, and thermodynamics.
Qus 8. How does conservation of energy relate to thermodynamics?
The Law of Conservation of Energy forms the basis of the First Law of Thermodynamics. It states that energy in a system can change forms, such as from heat to work, but the total energy remains constant. In thermodynamics, this principle helps explain energy transfer between systems and surroundings. It is fundamental to analyzing engines, refrigerators, and all heat-related processes.
Qus 9. How does an interactive simulator help in understanding conservation of energy?
Simulators allow learners to visualize energy transformations, adjust variables, and observe real-time changes, making abstract concepts easier to understand.